 Sodium’s electron configuration is 1s 22s 22p 63s 1. Sodium has an electronegativity of 0.93 (Pauling Scale). Furthermore, sodium will readily bond with any element that needs a single electron. This element is in the alkali metals family, and it has only one valence electron. Sodium, atomic symbol Na, has an atomic number of eleven, and lies in group 1 on the periodic table, below lithium and to the left of magnesium. Large quantities of sodium were disposed of in a lake in an explosive fashion after WWII ( ).Researchers are trying to learn about different the sodium content of different stars, in order to predict their evolution.So sodium’s elemental symbol ‘ Na’ is derived from this word. Latin for sodium carbonate is ‘ natrium’.A knife can cut through sodium metal quite easily.The element sodium accounts for 2.6% of the Earth’s crust.Sodium density is lower than that of water’s.Interestingly, 0.15% of your body is sodium.Sodium is the sixth most abundant element on Earth.That is to say, Roman soldiers were often paid their allowance in salt, which was considered quite valuable in their time. The monthly allowance of Roman soldiers was referred to as ‘ salarium’ – in which ‘ sal’ is Latin for salt. 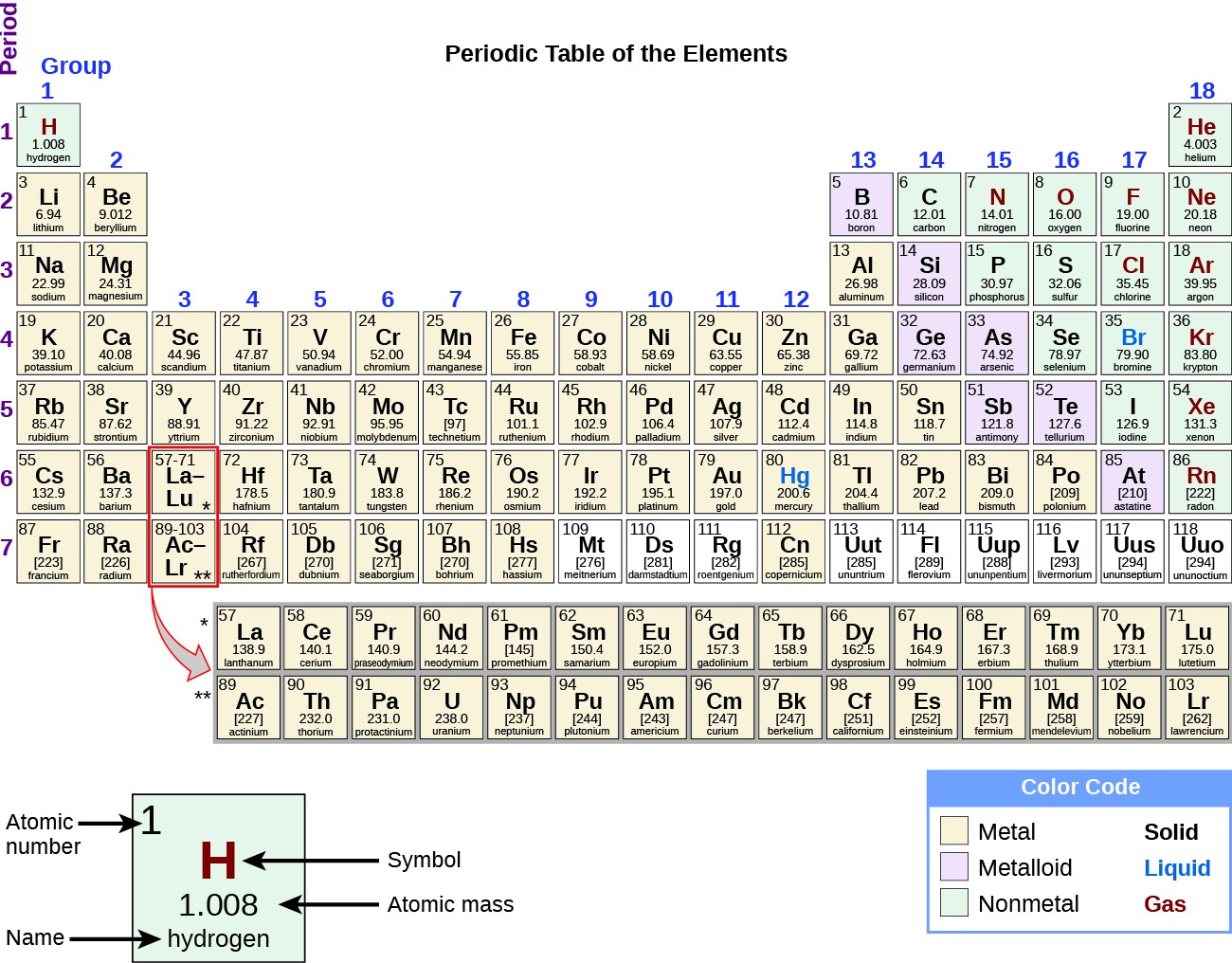
You might be familiar with table salt, which is NaCl (sodium chloride), a compound of sodium. It is one of the most reactive metals on the periodic table. Sodium, named after the English word ‘soda’, has compounds which are commonly used in our day-to-day lives. The element sodium is a soft, flammable, silvery-white metal. The electronic configuration of Sodium will be 1s2 2s2 2p6 3s1.The Element Sodium Introduction to Sodium How do you write the electron configuration for Sodium? The electronic configuration of Sodium will be 1s2 2s2 2p6 3s1. What is the electronic configuration of Sodium 11? What is the boiling Point of Sodium in Kelvin?īoiling Point of Sodium in Kelvin is 1156 K. Melting Point of Sodium in Kelvin is 370.87 K. What is the melting Point of Sodium in Kelvin? What is the boiling Point of Sodium?īoiling Point of Sodium is 1156 K. Sodium has 11 electrons out of which 1 valence electrons are present in the 3s1 outer orbitals of atom. How many valence electrons does a Sodium atom have? It is located in group 1 and period 3 in the modern periodic table. Sodium is the 11 element on the periodic table. Sodium is a chemical element with the symbol Na and atomic number 11. What is the position of Sodium in the Periodic Table? Sodium is a chemical element with symbol Na and atomic number 11. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets. The abbreviated electronic configuration of Sodium is 3s1. What is the abbreviated electronic configuration of Sodium? The electronic configuration of Sodium is 1s2 2s2 2p6 3s1. 
What is the electronic configuration of Sodium? Sodium Thermal Properties - Enthalpies and thermodynamics Optical Properties of Sodium Refractive IndexĪcoustic Properties of Sodium Speed of Sound Sodium Heat and Conduction Properties Thermal Conductivity Refer to table below for the Electrical properties ofSodium Electrical Conductivity Hardness of Sodium - Tests to Measure of Hardness of Element Mohs Hardness Refer to below table for Sodium Physical Properties DensityĠ.968 g/cm3(when liquid at m.p density is $0.927 g/cm3) 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
